How blinking affects the tear film lipid layer in the eye
Dry eye syndrome is a painful condition affecting millions of people throughout the world and is caused by the evaporation of tear fluid from the eye surface. What causes this evaporation is the focus of a PRACE project led by Professor Ilpo Vattulainen and a team at the University of Helsinki, with the work concentrating on the lipid molecules on the eye surface and how their behaviour is affected by blinking.
Dry eye syndrome (DES) is one of the most common diseases of the eye, estimated to affect more than 400 million people around the world. The condition is caused by excessive evaporation of tear fluid from the surface of the eyes, leaving them dry and scratchy and often causing blurred vision. It can be exacerbated by prolonged interaction with computer screens and in air-conditioned rooms where the atmosphere is very dry.
It is not clear why some people suffer with DES and others do not. Treatments are available in the form of eye drops, but these need to be applied regularly to ease the recurring symptoms. If left untreated, the condition can in rare cases lead to blindness.
What is known about DES is that the excessive evaporation of tear fluid is linked to the impaired function of the tear film lipid layer (TFLL) that covers the watery film over the eyes. The TFLL keeps the eyes moist by providing this surface layer of fluid with resistance to evaporation. How it does this and why it sometimes fails has been the focus of a PRACE project led by Professor Ilpo Vattulainen and his team at the University of Helsinki.

Vattulainen specialises in the field of biological physics, using theoretical and computational techniques to explore the underlying physical principles that govern the behaviour of biological systems. With a particular focus on lipid membranes and the ways that they modulate key biological processes, his group has been applying its knowledge and methods to try and understand the mechanisms behind the evaporation resistance of the TFLL.
Previous simulation work by the group had shown how wax esters, lipid molecules that are present at the surface of the eye in large amounts, are able to extensively slow down water evaporation. However, these simulations looked at the TFLL in a state of equilibrium, which is not a realistic representation of what occurs in the eye, as Vattulainen explains: “When looking at a computer display, you blink your eyes every 5-10 seconds. Each time this happens, the lipid layers on the surface of your eyes are compressed and then gradually expand afterwards.
“The question we wanted to answer is how this process of compression and expansion, which creates a constant state of non-equilibrium, affects the reorganisation of the lipid layers, and how this in turn is linked to water evaporation.”
The team simulated this non-equilibrium process thoroughly, performing many repeats under various conditions. “We looked at how evaporation changed in systems with wax esters compared to those without, and also did the same for other similar lipids such as cholesteryl esters. Lipid by lipid, we checked the importance of these for the stability of the lipid layers at the surface of the eye, checking their importance in retarding water evaporation while trying to simulate the layers in as realistic conditions as possible.”
These atom-scale molecular dynamics simulations have provided the first-ever atomistic view into the non-equilibrium properties of the TFLL. The team discovered that through blinking, cholesteryl esters and wax esters in the non-polar lipid bulk form multilayered crystal-like structures that act as an evaporation resistant barrier. Polar lipids also settle against the water, spreading the TFLL over the layer of moisture.
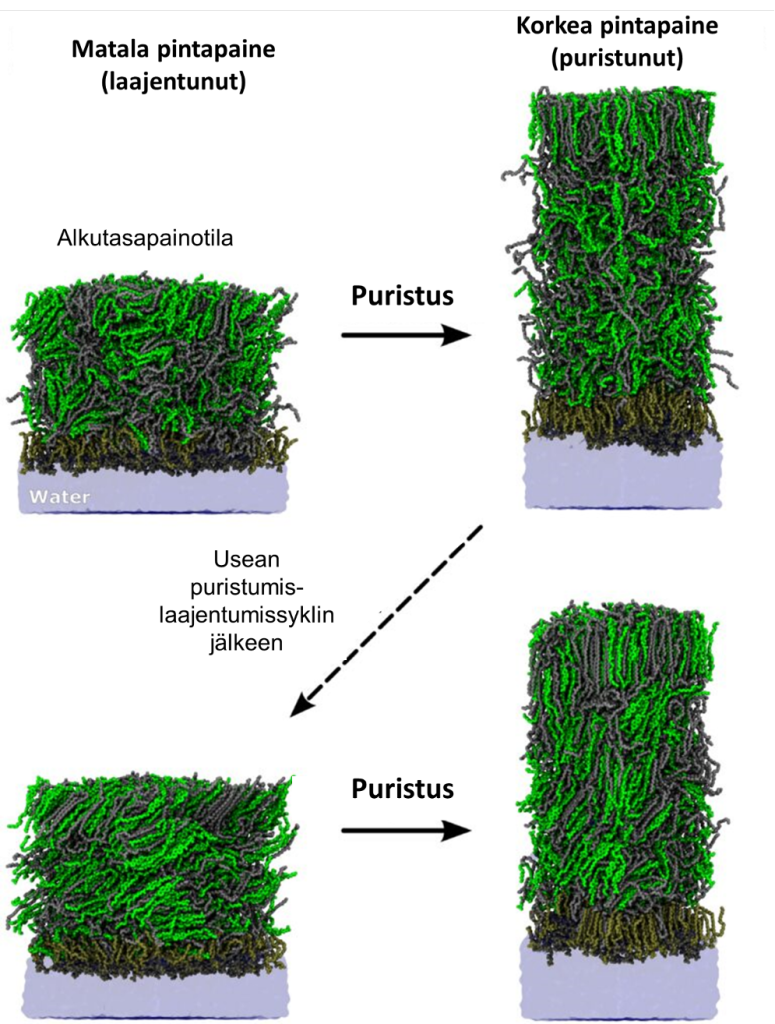
It was also discovered that while in a disordered lipid structure, water molecules have more ways to escape from the water phase and travel into the air, the highly-packed, crystal-like structure of the non-polar lipid components acts as an effective barrier, retarding water evaporation. The structure of the bulk phase of the TFLL is controlled by lipid composition and thermodynamic conditions.
“The big piece of news to emerge from this project is that the non-equilibrium behaviour is quite different to the equilibrium behaviour that occurs without any blinking,” says Vattulainen. “In a practical sense, this could lead to eye drops for DES that contain wax esters, which they don’t at the moment. This would mean that the drops would last longer and wouldn’t need to be applied every hour.”
A number of further lines of research could be explored to follow up this project. While the TFLLs studied simulated ranged from 20-30 nanometres in thickness, they tend to be closer to 100 nanometres in reality. This would however require much larger computing resources. As well as this, the simulations did not take into account any of the proteins that are present in real TFLLs. “A number of surfactant proteins that are known to exist in the lung, including those that enable oxygen permeation into the blood, are also found in the TFLL,” says Vattulainen. “So far, no experimental work or simulations have managed to explain the presence of these proteins in the eye.”
Vattulainen and his Helsinki team are confident that progress in understanding DES will continue thanks to LUMI, a new petascale machine under construction in Kajaani, Finland, of which Finland will own 25% of the computing capacity. “But when we applied to PRACE for this project, our own computing resources were quite small,” he says, “so we were extremely happy that PRACE decided to support us.”
Resources awarded:
This project was awarded 67 000 000 core hours on Joliot-Curie in Rome, hosted by GENCI/CEA, France.
Details of the original article:
- This article was originally published in PRACE Digest 2021 and it can be found on PRACE website.
- The original article has been written by Harry O’Neill of Insight Media Group
Writer: Harry O’Neill
